Local market registration requires a full understanding of local regulations, policy changes and industry trends. Brilliantmed provides comprehensive local registration support, covering:
· Regulatory compliance consultation: Based on the changes in regulations, we provide enterprises with local market registration strategies and interpretation of regulatory requirements.
· Registration document preparation and review: Ensure that the application materials meet the technical requirements of local regulatory agencies and improve the approval success rate.
· Registration project management: From application to approval, track and coordinate communication with regulatory agencies throughout the process to ensure efficient progress of the process.
· Government Relations and Communication Support: Work closely with regulatory agencies (such as NMPA, FDA, EMA, HSA, etc.) to help companies deal with challenges in the review process.
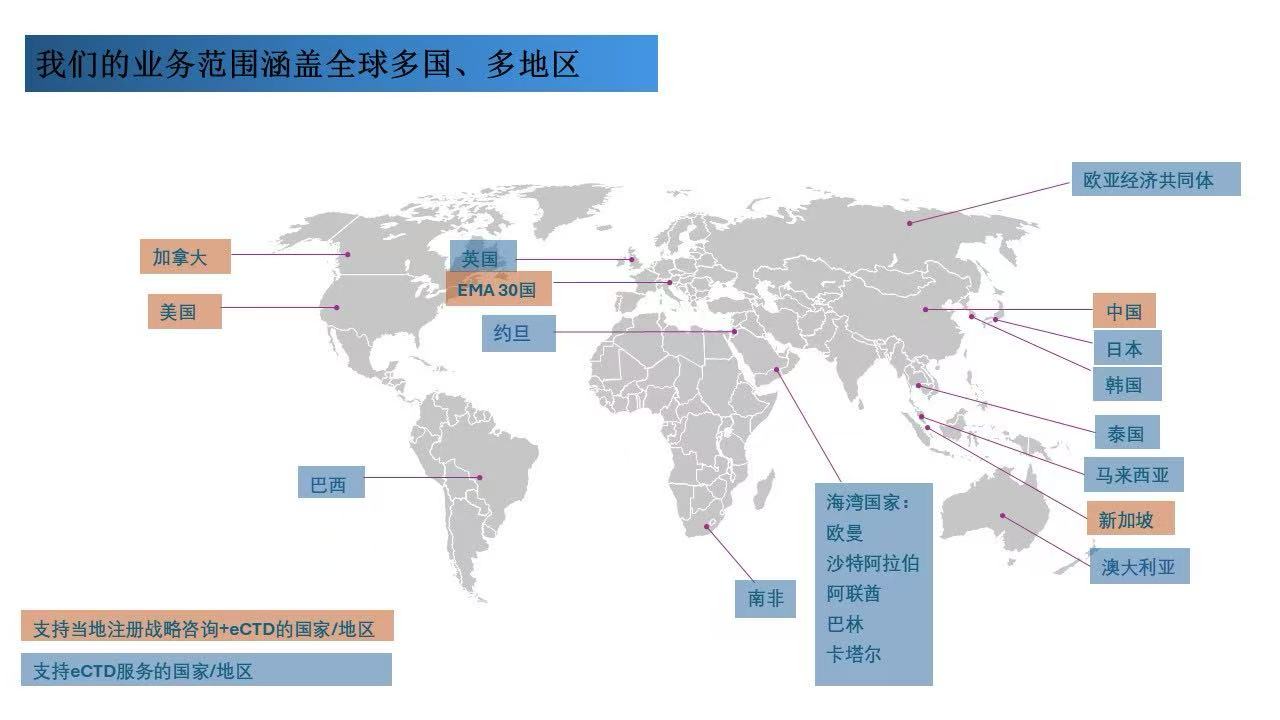
We are deeply engaged in the local market and provide customers with accurate and efficient registration support, helping our products get approval smoothly.
Looking ahead, we will continue to strengthen technological innovation and service upgrading, promote the healthy development of the pharmaceutical industry, and create greater value for customers. We look forward to cooperating with global customers to create brilliance together.
 Room 508, Building 8, Zhongke E-commerce Valley, No. 20 Guangde Street, Beijing Economic and Technological Development Area, Beijing City
Room 508, Building 8, Zhongke E-commerce Valley, No. 20 Guangde Street, Beijing Economic and Technological Development Area, Beijing City
 +86 15001236015
+86 15001236015
 Sophia.huang@brilliantmed.cn
Sophia.huang@brilliantmed.cn